Clinical Reasoning: A 64-year-old man with painful, unilateral external ophthalmoplegia
M. Tariq Bhatti. Neurology. 2010; 75 (8)
RESIDENT AND FELLOW SECTION
SECTION 1
A 64-year-old man was referred for evaluation of double vision in March 2009. Four months prior, he experienced daily pain in the region of the right forehead and right eye. Two months later, he noticed constant binocular, vertical double vision. Evaluation by an outside ophthalmologist resulted in the diagnosis of a right 4th cranial nerve (CN) palsy, a normal cranial and orbital MRI study with contrast, and an unremarkable laboratory evaluation. One month later, his right eye began “turning in,” and within a few days he was unable to abduct the eye. Several weeks later, the right eyelid began to droop and progressively worsened over the next several days to complete closure of the eye.
His past medical history was notable for arterial hypertension, depression, rheumatoid arthritis, gastroesophageal reflux disease, nephrolithiasis, squamous cell carcinoma of the forehead, and a precancerous melanoma of the left ear.
When he was seen in the neuro-ophthalmology clinic, visual acuity was 20/20 in each eye. Color vision was intact in each eye. The right pupil was 2 mm larger than the left and was nonreactive to light or near effort. There was no relative afferent pupillary defect (RAPD). Dilated fundus examination was normal in each eye. Eye movements of the right eye were limited in all directions and there was complete right upper eyelid ptosis (figure 1). Corneal sensation of the right eye was absent and there was numbness over the right forehead. The remainder of the cranial nerve examination was normal.
Figure 1 Nine cardinal positions of eye movements
There is limited movement of the right eye in all directions of gaze. There was no intorsion of the right eye on attempted downgaze. The right eyelid is manually elevated because of the complete right eyelid ptosis (center, middle panel). Permission obtained from patient.
Questions for consideration:
1. What is the clinical presentation?
2. Where does the lesion localize?
SECTION 2
This 64-year-old-man presented with a painful, complete external ophthalmoplegia of the right eye. His history and examination were consistent with sequential CN 5, 4, 6, and 3 palsies. To ascribe a lesion in one anatomic location to cause such a clinical picture would require the lesion to be in the superior orbital fissure, cavernous sinus, or both. Although it is possible a more proximal lesion (i.e., as the CNs exit the brainstem) could result in CN 3, 4, 5, and 6 paresis, the fact that the patient did not have any meningeal or brainstem signs or symptoms makes it unlikely. Because CNs 3, 4, and 6 pass through both the cavernous sinus and superior orbital fissure, a lesion in either of these 2 anatomic regions is often difficult to clinically distinguish, resulting in the term sphenocavernous syndrome. However, careful assessment of CN 5 function can aid in differentiating involvement between a lesion in the cavernous sinus or superior orbital fissure. The second division of CN 5 exits the cavernous sinus into the foramen rotundum; therefore, it is not involved in a superior orbital fissure syndrome. Even though a normal clinical examination of the second division of CN 5 favors a lesion in the superior orbital fissure, this does not completely exclude the possibility of cavernous sinus involvement in the setting of a progressive disease process. Evidence of an optic neuropathy (CN 2 palsy)—as manifested by visual loss, impaired color vision, and a RAPD—in the presence of an ipsilateral external ophthalmoplegia indicates an orbital apex syndrome.
Question for consideration:
1. What is the differential diagnosis?
SECTION 3
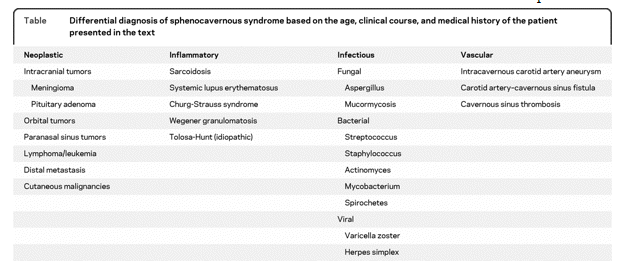
The differential diagnosis of a sphenocavernous syndrome is extensive and can be the result of systemic disease, metastatic disease, or primary lesions arising from the head and neck, orbit, cranium, paranasal sinuses, or nasopharynx (table). Age, prior medical history, and race of the patient are important determinants in formulating a differential diagnosis. The onset of symptoms and the presence of pain are also very important factors. An acute onset of symptoms would favor a vascular event or an infectious process, while a progressive course would raise the possibility of a neoplastic process, especially given this patient’s prior history of malignancy. Ocular or facial pain indicates CN 5 involvement and can be the result of a compressive, inflammatory, or infiltrative condition. The Tolosa-Hunt syndrome is an idiopathic, inflammatory condition involving the cavernous sinus, superior orbital fissure, or orbital apex. It is a diagnosis of exclusion and a thorough evaluation should be performed to exclude more specific etiologies.1
Question for consideration:
1. What studies and/or tests do you want to perform?
SECTION 4
A cranial and orbital MRI with contrast was performed and demonstrated an enhancing mass in the right superior orbital fissure, anterior cavernous sinus, and right supraorbital nerve (figure 2).
Figure 2 MRI
(A) Axial, postgadolinium, fat-suppressed MRI demonstrates an enhancing lesion of the right superior orbital fissure and anterior cavernous sinus (arrow).
(B) Coronal, postgadolinium, fat-suppressed MRI shows enlargement and enhancement of the right supraorbital nerve (arrow).
A right supraorbital nerve biopsy was performed. The histopathologic specimen was consistent with perineural invasion (PNI) from cutaneous squamous cell carcinoma (SCC) (figure 3).
Figure 3 Histopathology
Histopathologic specimen of right supraorbital nerve biopsy showing neoplastic cells scattered among the supraorbital nerve fibers. (A, Hematoxylin-eosin stain; B, keratin stain.).
DISCUSSION
Neoplastic cells can metastasize throughout the body by several different mechanisms, including the often overlooked mode of PNI. PNI is defined by the spread of neoplastic cells via the nerve-nerve sheath complex. Carter et al.2 showed, in pathologic specimens of 65 patients with SCC of the head and neck, varying degrees of demyelination, axonal degeneration, and segmental infarction of the infiltrated nerves, possibly due to hypoxic/anoxic injury.
PNI has been reported to occur in approximately 5% of cutaneous SSC cases.3 The most common CNs involved are the 5th (second and third division) and 7th. CN 5 involvement can be misinterpreted as trigeminal neuralgia in some cases, and in other cases the dysesthesia and hypesthesia can be vague, nonspecific, and nonlocalizing. Facial muscle weakness is indicative of CN 7 involvement.4 PNI from cutaneous SCC resulting in ophthalmologic and orbital involvement most commonly arises from the forehead and eyebrow regions.
The radiologic study of choice in cases suspected of PNI is a cranial and orbital MRI with contrast and fat suppression. MRI findings include an enhancing mass, enlargement and enhancement of CNs, obliteration of normal fat planes, and enlargement of foramina or canals.5
The diagnosis of PNI can be challenging for several reasons, including negative neuroimaging in as many as 50% of cases,6 diagnostic mimickers, vague symptomatology, incomplete or absent medical history of a cutaneous malignancy, and an extended interval from the time of the primary diagnosis of cutaneous malignancy to onset of clinical manifestations. Fine-needle aspiration biopsy, open surgical biopsy, or peripheral nerve biopsy can often establish the diagnosis.
The prognosis of PNI of cutaneous SCC is poor. The 5-year survival rate of cutaneous malignancies with PNI is approximately 50%.3 Definitive radiation therapy, preferably conformal or intensity-modulated radiation therapy, is recommended for nonresectable cases of PNI.3 Orbital exenteration may be warranted for some patients with disease confined to the orbit.
Follow-up.
The patient received a total dose of 5,600 cGy delivered by fractionated intensity modulated radiation therapy. At his 6-month follow-up, the neuro-ophthalmic examination was essentially unchanged.
DISCLOSURE Dr. Bhatti has served on speakers’ bureaus for and received speaker honoraria from EMD Serono, Inc., Pfizer Inc., Bayer Schering Pharma, and Novartis.
REFERENCES.
1. Kline LB, Hoyt WF. The Tolosa-Hunt syndrome. J Neurol Neurosurg Psychiatry 2001;71:577–582.
2. Carter RL, Foster CS, Dinsdale EA, Pittam MR. Perineural spread by squamous carcinomas of the head and neck: a morphological study using antiaxonal and antimyelin monoclonal antibodies. J Clin Pathol1983;36:269–275.
3. Mendenhall WM, Amdur RJ, Hinerman RW, et al. Skin cancer of the head and neck with perineural invasion. Am J Clin Oncol 2007;30:93–96.
4. Scurry WC, Jr, Isaacson JE, Fedok FG. New-onset facial paralysis and undiagnosed recurrence of cutaneous malignancy: evaluation and management. Am J Otolaryngol 2006;27:139–142.
5, Gandhi D, Gujar S, Mukherji SK. Magnetic resonance imaging of perineural spread of head and neck malignancies. Top Magn Reson Imaging 2004;15:79–85.
6. Williams LS, Mancuso AA, Mendenhall WM. Perineural spread of cutaneous squamous and basal cell carcinoma: CT and MR detection and its impact on patient management and prognosis. Int J Radiat Oncol Biol Phys 2001;49:1061–1069.